We are trying to develop a summary of what needs to be done to develop an adequate basis for the scientific analysis of post-medieval vessel glass. This includes the type of glass on this web site. The common methods of glass analysis do not work well for glass of this period, since we need to analyse both excavated and complete examples, ideally using the same technique. Also, there is strong evidence that these glasses are not homogeneous, so that a small sample taken from one part of the glass may be very different from that in another part. So we need different methods to find out what chemicals are present in the glass. The fluorescence technique described next might be one way of doing it, but to be sure we first need to work out what results we need. But finding the chemical make-up is not the whole story, simply knowing what chemicals are there does not take you very much further forward unless you can use this information to help you answer important questions. We would be delighted to receive views and contributions to this debate. Click here to download the current summary document and please email us with any comments you have so we can incorporate these
Colin & Sue Brain
This short report documents examples of fluorescence responses at certain combinations of excitation and emission wavelengths and discusses the attribution of these to specific components in the glass. Examples are selected to demonstrate specific points from forty-six analysis runs recently undertaken. In virtually all cases similar responses occurred on other analysis runs, but the features exhibited were less clear-cut. The use of measured glass fluorescence in this way to identify the component ions present in glass is a relatively novel approach. The aim of this report is to briefly summarise the evidence available to link the measured fluorescence of the glass to important components used in its manufacture. This then provides a basis for drawing conclusions about the glass, based on the presence or absence of these components.
The bulk of the glass came from Templeogue, near Dublin and appears to form a stylistic group, probably manufactured over a relative short period of time, possibly less than ten years. All the major fragments apparently belonging to this group were analysed. Analysis runs were also carried out on similar glasses from Dublin Castle, Port Royal (Jamaica) and Wells Museum. There is documentary evidence supporting the export of glass from Ireland to Port Royal, but no evidence has yet been identified to support an Irish origin for the Wells glass. The glasses analysed may all fall within the period 1678-1688. I am grateful to The National Museum of Ireland, the Board of Works, Texas A&M University, Jamaica National Heritage Trust, the Institute of Nautical Archaeology, Wells Museum, Peter Frances, Donny Hamilton and Chris Hawkes for the opportunity to make these measurements.
Glass fluorescence to UV/visible light was measured using a modified Aminco Bowman fluorescence spectrometer. The fluorescence excitation was from a Xenon arc-lamp source and was dispersed by a grating monochromator, giving an excitation wavelength range of 230 to 380 nm. Measurements were made at 10 nm intervals under computer control, with the wavelength measurements being electrically calibrated at the start of each day’s measurements. Fluorescence from the glass was also dispersed by a grating monochromator and sensed with a photomultiplier. The spectrometer utilises an excitation light by-pass and a chopper wheel to enable true ratio-metric measurements. The output from the photomultiplier is fed to the computer via a 12-bit D/A converter with the output wave-form being synchronised by software. The output range scanned was 330 to 630 nm (UV/violet-orange/red) at 10 nm intervals, except that no measurements were made within 40 nm of the excitation wavelength. The output data was post-processed to remove the effect of surface reflections. Both emission and excitation monochromator positions have been calibrated against the standard emission ‘lines’ for a mercury lamp.
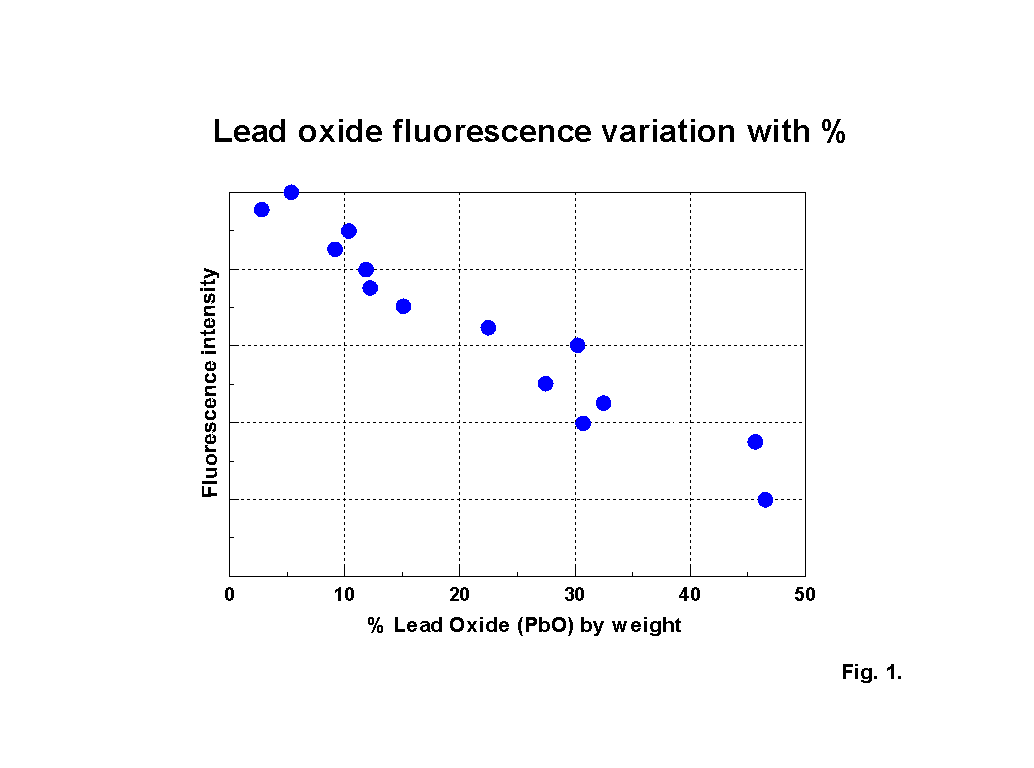
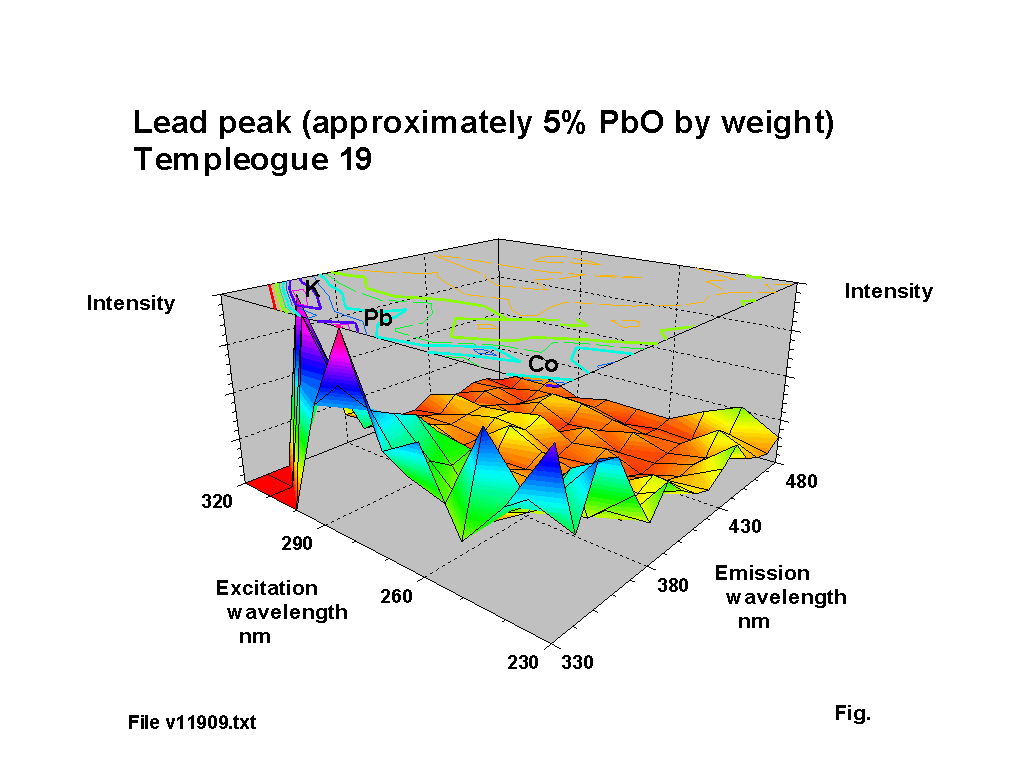
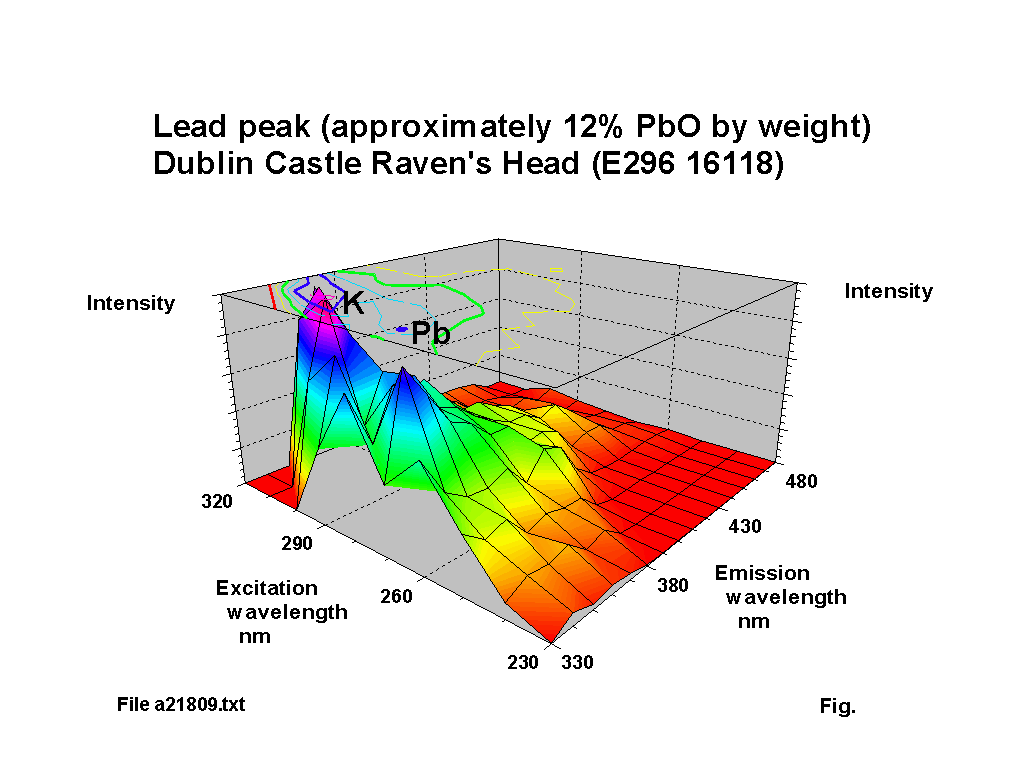
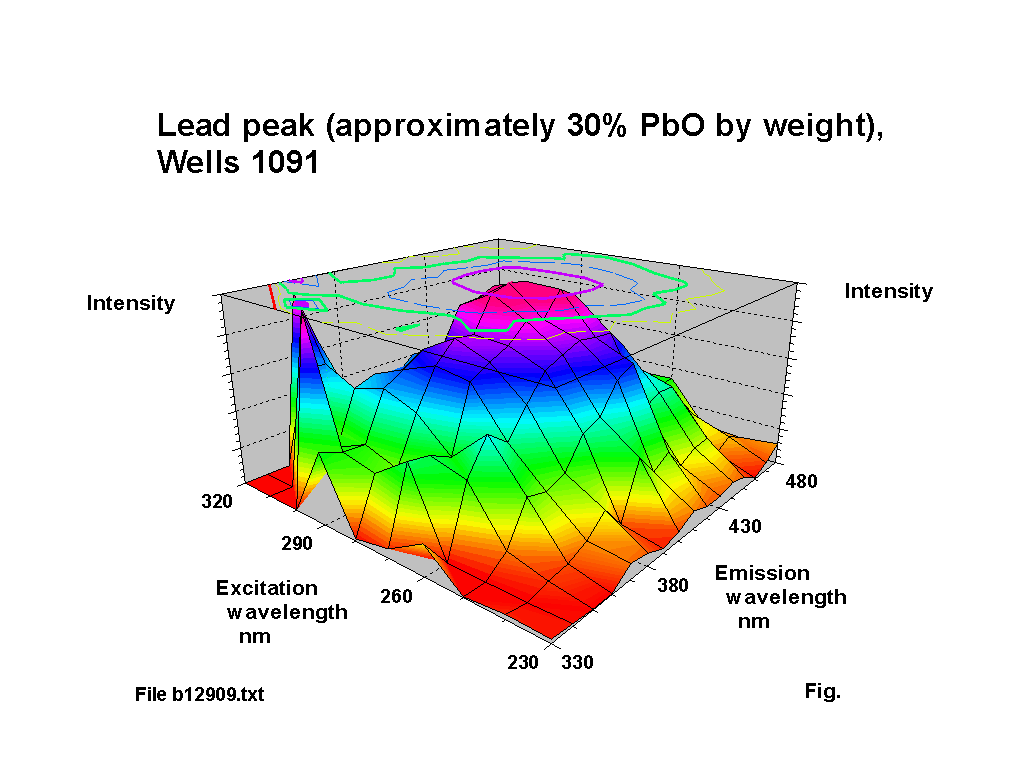
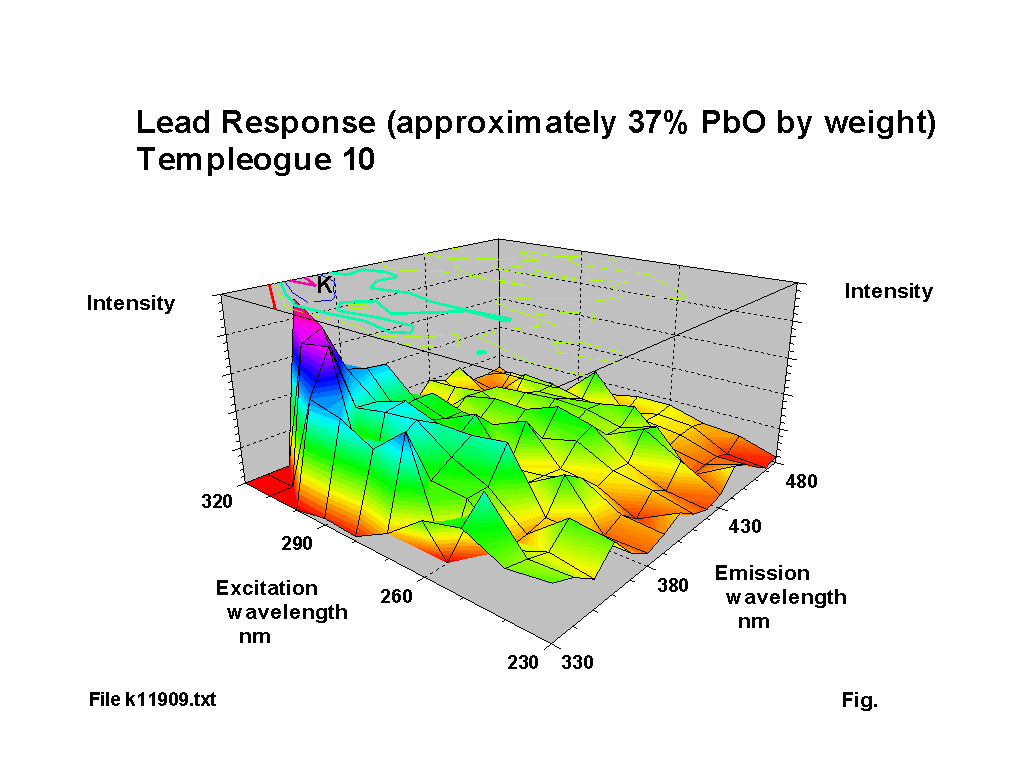
The blue fluorescence of lead glass under ‘Hg’ UV lamps is one of the most commonly known responses. The fluorescence peak location for lead oxide shifts to a reducing excitation wavelength and an increasing emission wavelength NOTE: subsequent analysis using calibration samples kindly provided by th Corning Museum of Glass has indicated that the variation is more complex than the simple trend suggested here; a paper including some of this new work was presented to the XIX International Congress on Glass, Edinburgh in July 2001 and a copy of the paper is available here (184KB). as the lead content increases, up to about 30% PbO by weight. Above this level of lead content the response becomes diffuse. Figures 2-5 show characteristic outputs over a range of lead contents. The % lead content of these pieces was estimated from their measured specific gravities. Figure 1 shows a graph, after [1], of the variation of fluorescence intensity with % PbO in commercial flint glasses. The Dublin glass results seem to follow this trend. Figures 2-5 also show the peaks due to potash and calcium.

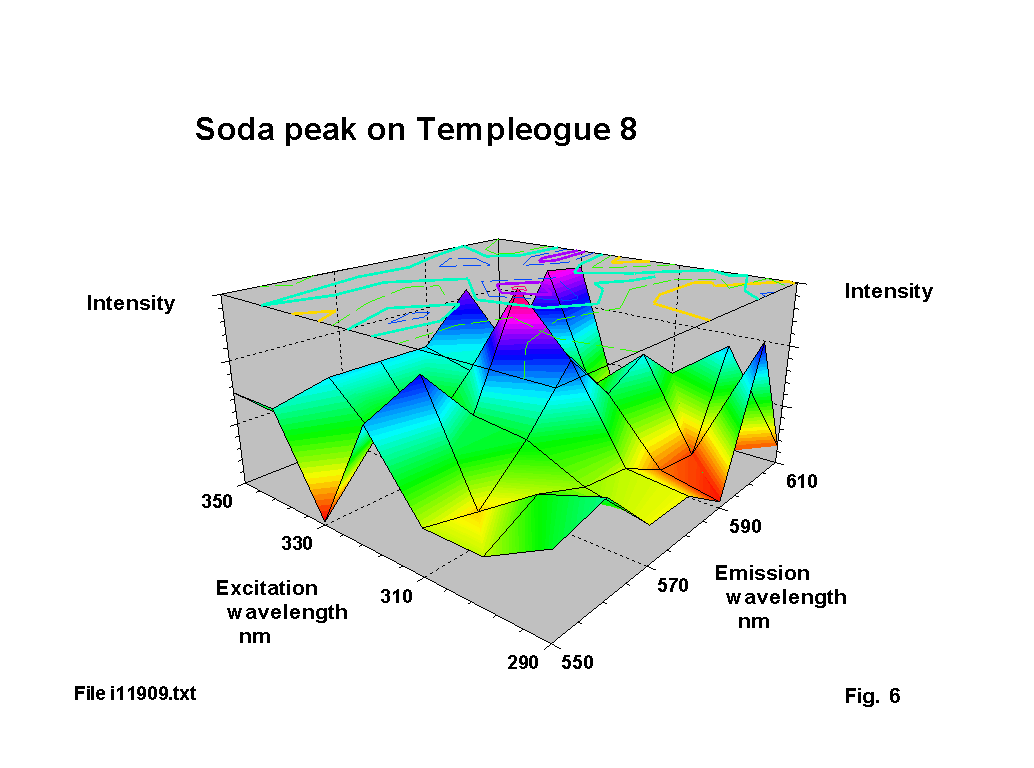
The yellow emission of soda is often demonstrated in flame tests. In glass specimens it is much more difficult to detect. There are two reasons for this. First the absorbtion coefficient of soda at the excitation wavelengths is relatively low. Second there may also be pronounced absorbtion of the excitation light by iron oxide and of the emitted light be cobalt or manganese oxide. Figure 6 shows the small soda peak measured on one of the glasses. The standard emission line for sodium (as a low pressure gas) is at 589 nm, with a corresponding absorbtion line at 330 nm. These match almost exactly the peak position on figure 6. However, not all soda glasses show this fluorescence and work on calibration samples show that there are a number of different possible soda fluorecence patterns. This is most likely to be due to variations in the optical basicity of the glass mix.
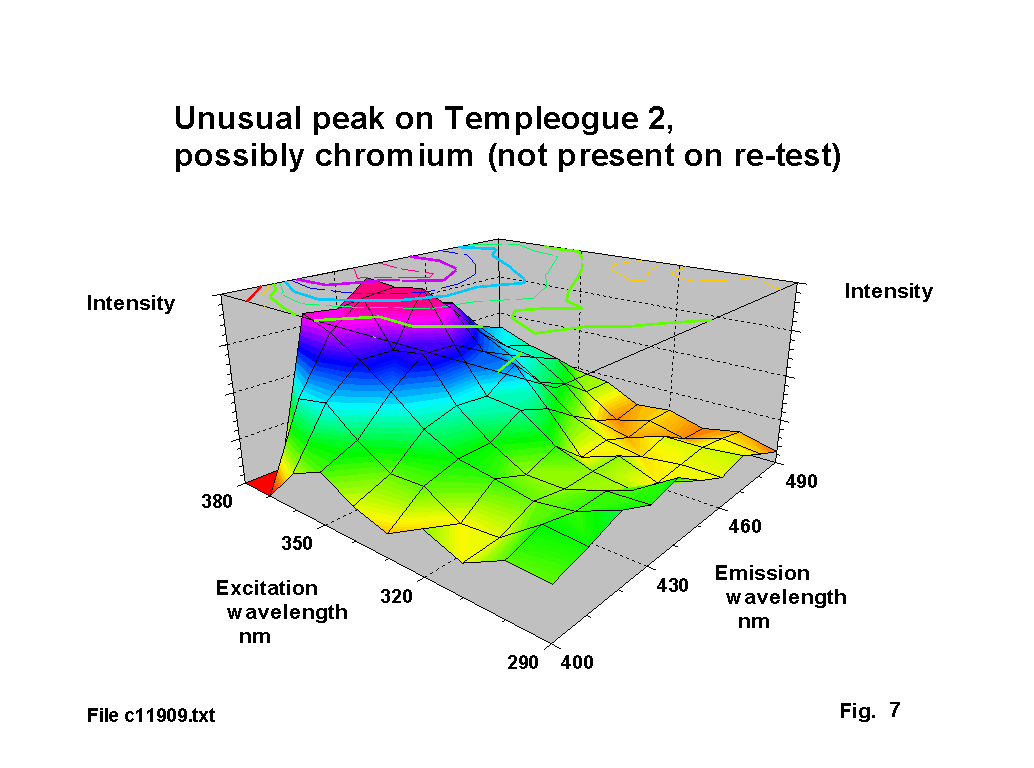
Chromium is an unusual ingredient for glass. It is sometimes used to achieve a green tint and only appears in one run on one glass. In this case it is almost certainly an impurity. A number of glasses of this period have small black specks and chromium is the most likely cause of these. The peak is shown in figure 7. Standard chromium emission lines are in the range of 425 to 429 nm, with absorbtion lines in the range 358-361 nm, which are in fair agreement with the peak position in this figure. Wong & Angel [4] record an absorbtion peak for the Cr6 ion at 366 nm, but this is linked to glass prepared under oxidising conditions. Watts [6] notes the presence of chromium in a Ravenscroft sealed stem fragment.
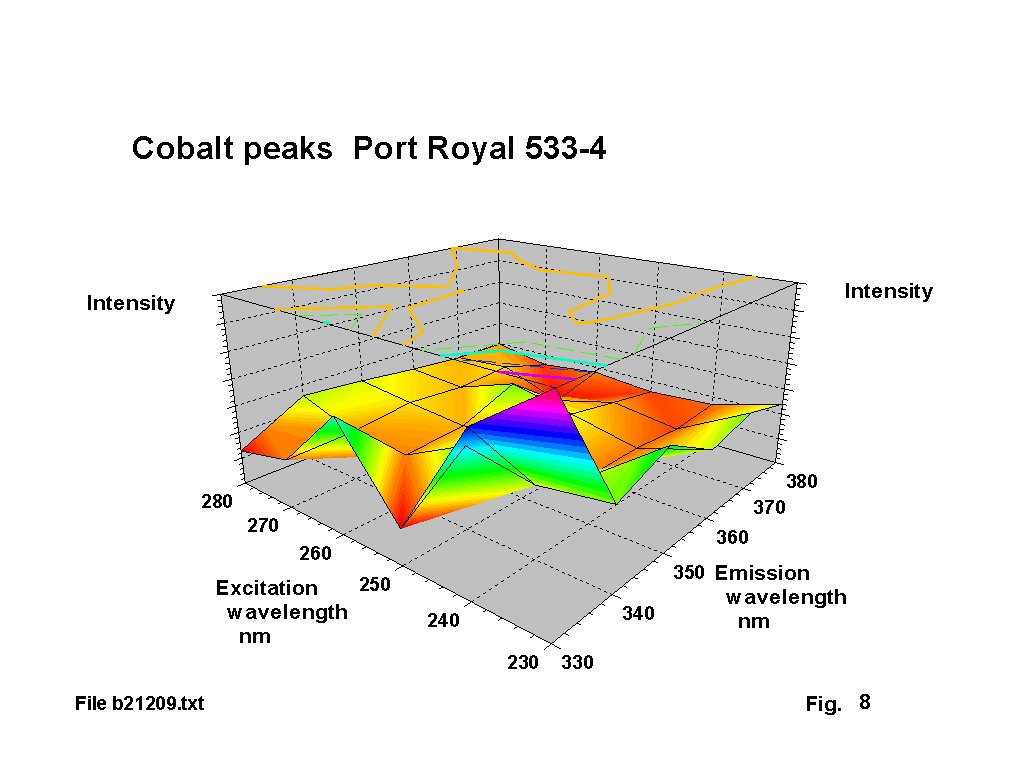
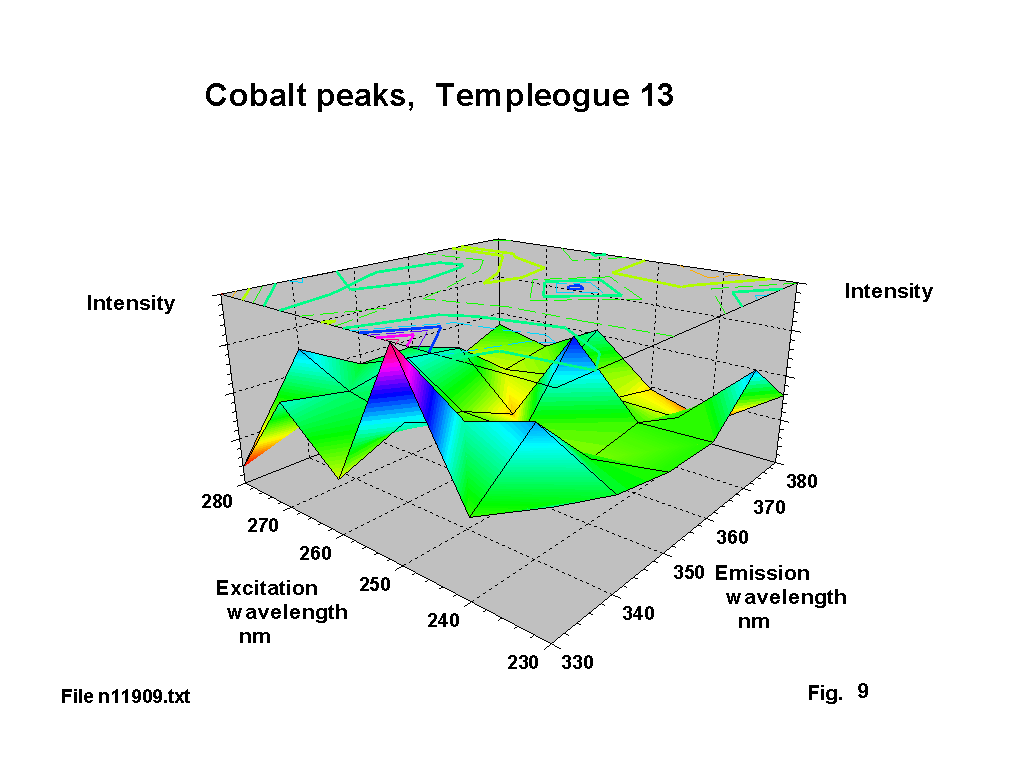
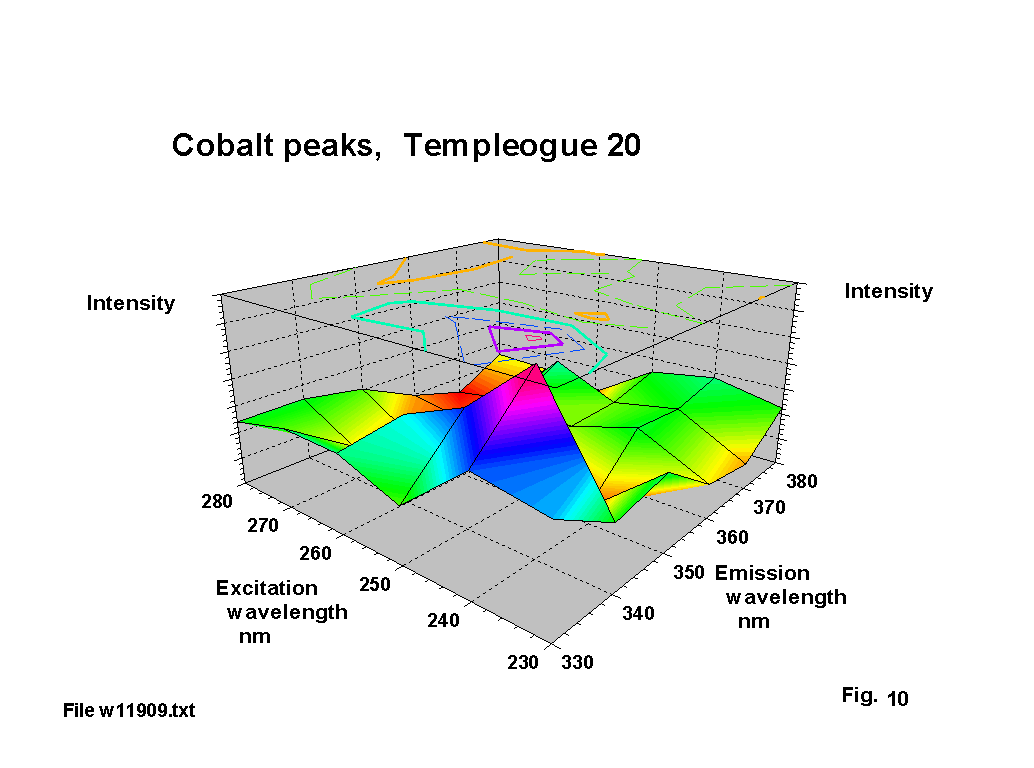
Cobalt seems to have been used deliberately as a decolouriser in a number of these glasses, and there is documentary evidence of cobalt-containing minerals being exported from Bristol to Ireland. The resultant blue tint is clearly visible. The standard emission lines for cobalt cover a number of ranges 339 - 358 nm, 375-389 nm and 399-412 nm. The corresponding absorbtion lines are in the range 239-252 nm. Figures 8,9 & 10 show a number of single and double peak responses in these ranges. Weyl [2] suggests cobalt gives a brown to green fluorescence and that the Co2 ion has an absorbtion peak at about 540 nm. Fuxi [3] gives absorbtion peaks at 500 and 616 nm. These are the classic absorbtion peaks that give rise to ‘cobalt’ blue glass, but are not evident to any extent on the output graphs because there is usually very little that fluoresces in this region.
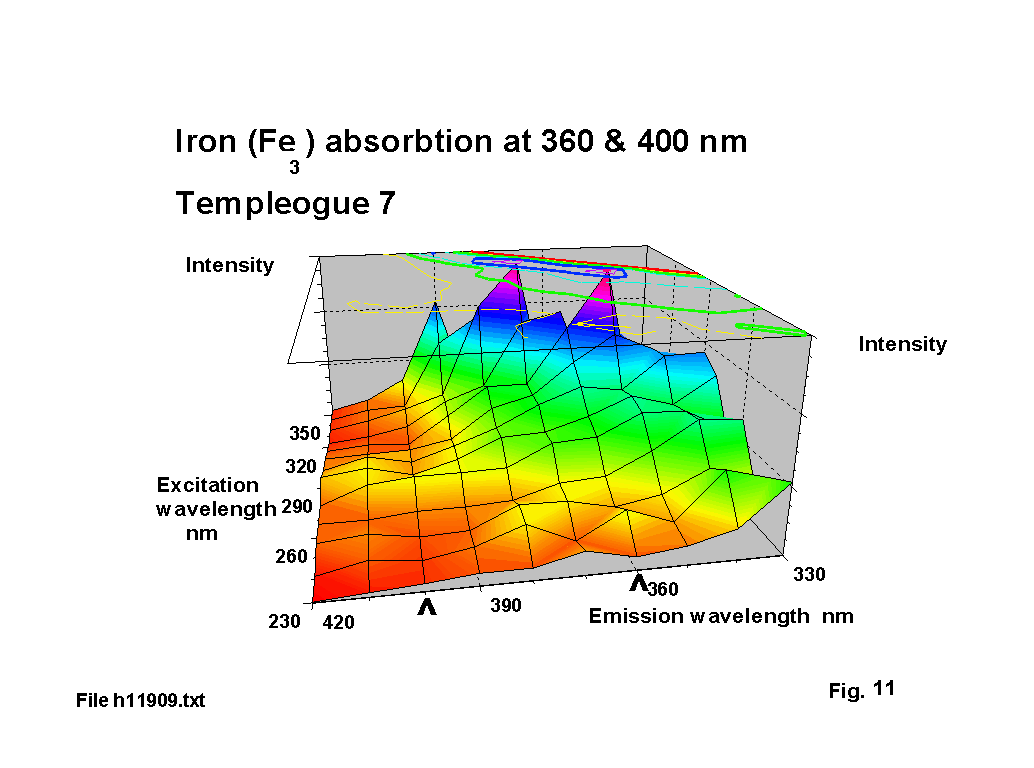
Iron does not fluoresce, but has marked absorbtion bands at visible wavelengths. Fuxi gives these as 380, 420 & 427 nm for the Fe3 ion in soda glass. Figure 11 illustrates absorbtion ‘valleys’ on one of the plots, centred on about 360 & 400 nm, which represents about a 20 nm shift, but this could simply reflect the effect of the changed glass matrix with significant lead content.
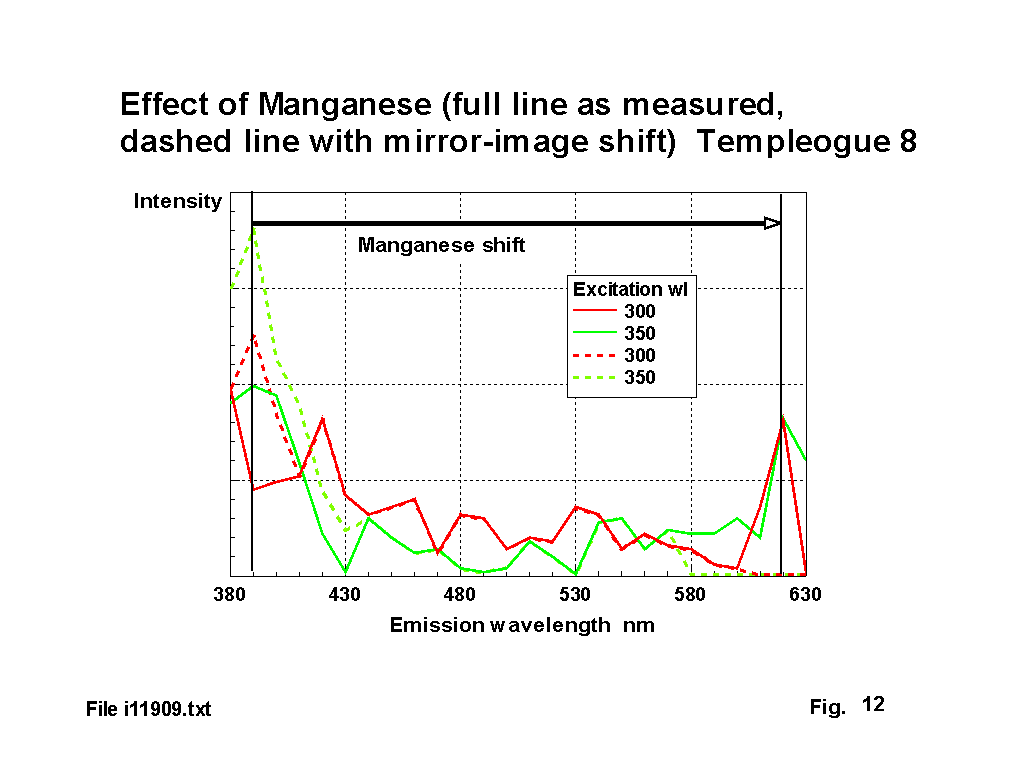
Manganese is one of the most difficult ions to deal with, because it absorbs and emits in the visible region. Previous workers have reported both green and red fluorescence and absorbtion peaks have been reported for the Mn2 ion at 355, 412, 418, 425, 430, 470 & 520 nm depending on the other glass constituents [2],[3],[4]. Fuxi reports Mn2 ion fluorescence at 526 & 606 nm. The Mn2 ion is only present in some glasses if they have been prepared under reducing conditions. The Mn3 ion has not been reported to fluoresce. Figure 12 illustrates the absorbtion - emission frequency shift for one glass, in this case from 390 nm to 620 nm.
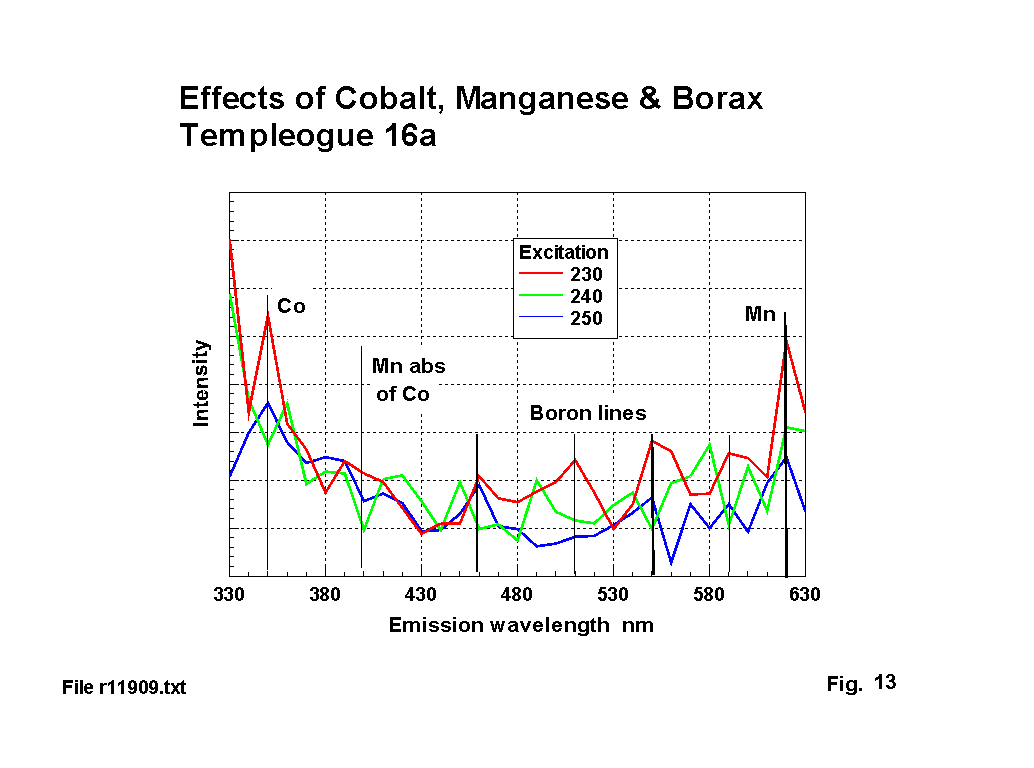
Figure 13 illustrates the combined effects of boron, manganese and cobalt. Both Radley & Grant [5] and Kreidl [1] give qualitative reports of boron fluorescence, but neither give measured peaks. Standard emission lines for Boron are: 450, 470, 490, 520, 545, 580, 600 nm, with absorbtion at 250 nm. The emission lines marked on figure 13 are: 460, 510, 550, & 590. This may represent a 10 nm shift, and the suppression of some lines. The peak absorbtion is also shifted by 10-20 nm.
These series of analysis runs represent an early application of this technique to archaeological glass. A number of fluorescence peaks have been identified and this report has suggested tentative allocations for them against ions present in the glasses. Evidence has been provided to support this allocation. Clearly the location of some absorbtion and emission peaks is influenced by the amounts and identities of other glass constituents. They are also influenced by the type of ion formed. This in turn depends on whether oxidising or reducing conditions were present in the batch at the time the glass is melted. Further work is required to improve the understanding of these peaks and their inter-relationships and to identify the locations of peaks due to other ions, such as calcium, aluminium, and magnesium that are likely to be present in small quantities in all these glasses. However, the allocation of peaks to these component ions is considered adequately supported to allow conclusions to be drawn about the glass making processes used.
[1] - Kreidl, N.J., Recent Studies on the Fluorescence of Glasses, Journal of the Optical Society of America, vol.35, No 4, April 1945, p.257.
[2] - Weyl, W.A., Coloured Glasses, published by Society of Glass Technology, 1951.
[3] - Fuxi, G., Optical and Spectroscopic Properties of Glass, Springer-Verlag
[4] - Wong, J. & Angel, C.A., Glass: Structures by Spectroscopy
[5] - Radley, J.A. & Grant, J., Fluorescence Analysis in Ultra-Violet Light, 4th edition, London, 1954, pp 145-6.
[6] - Watts, D.C., How did George Ravenscoft discover lead crystal, The Glass Circle, Vol 2, 1975, p.72.
Home | News | Contents | People | Identify | Science | Reference | Links
Copyright Colin & Sue Brain, 2000